KEYTRUDA + LENVIMA Clinical Data: Clear Cell and Non-Clear Cell RCC
Studied in the first-line setting across MSKCC risk groups
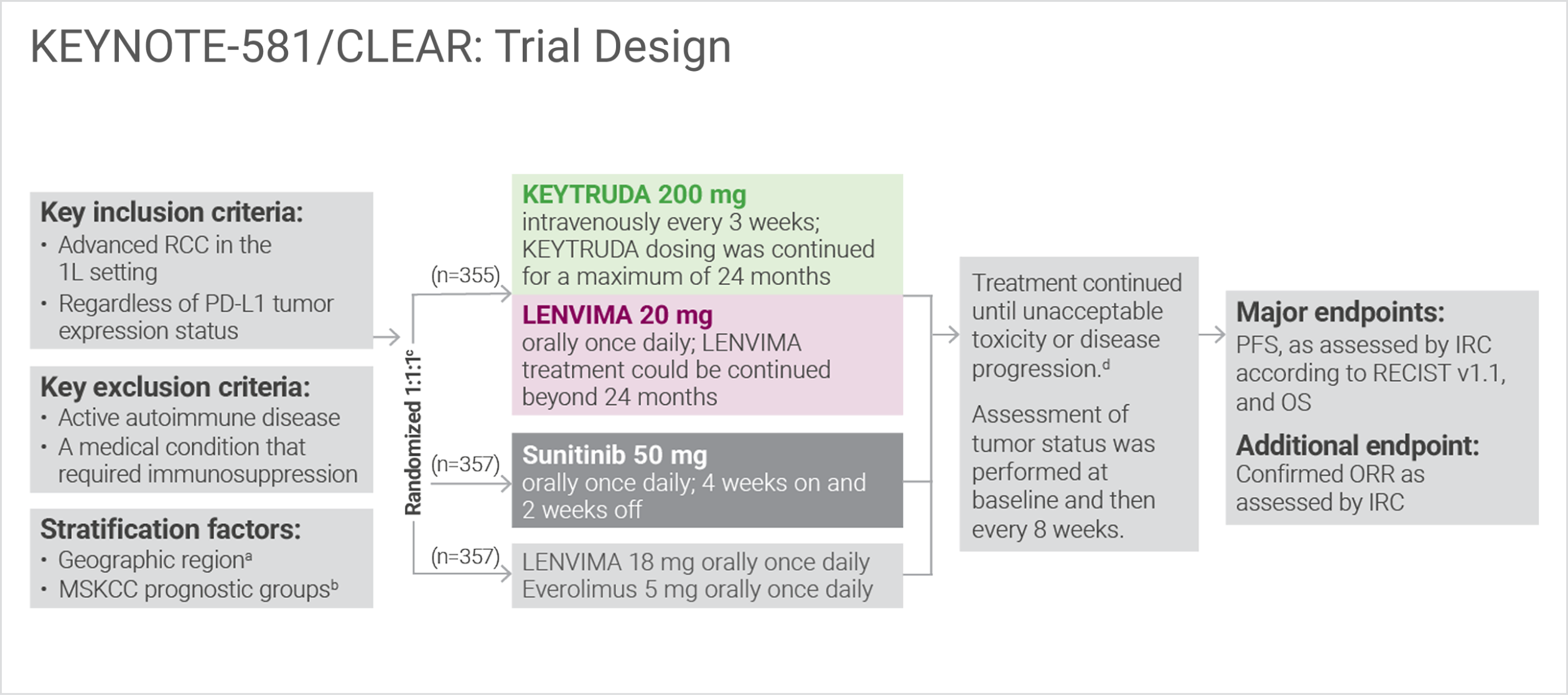
A multicenter, randomized, open-label, phase 3 trial with 1,069 patients1
North America vs Western Europe vs “Rest of the World.”
Randomization was stratified according to Memorial Sloan Kettering Cancer Center (MSKCC) prognostic risk groups: favorable vs intermediate vs poor.
Clinical data are presented from the KEYTRUDA + LENVIMA and sunitinib arms.
Administration of KEYTRUDA with LENVIMA was permitted beyond RECIST-defined disease progression, if the patient was clinically stable and considered by the investigator to be deriving clinical benefit.
1L = first-line; IRC = independent radiologic review committee; ORR = objective response rate; OS = overall survival; PD-L1 = programmed death ligand 1; PFS = progression-free survival; RCC = renal cell carcinoma; RECIST v1.1 = Response Evaluation Criteria In Solid Tumors v1.1.
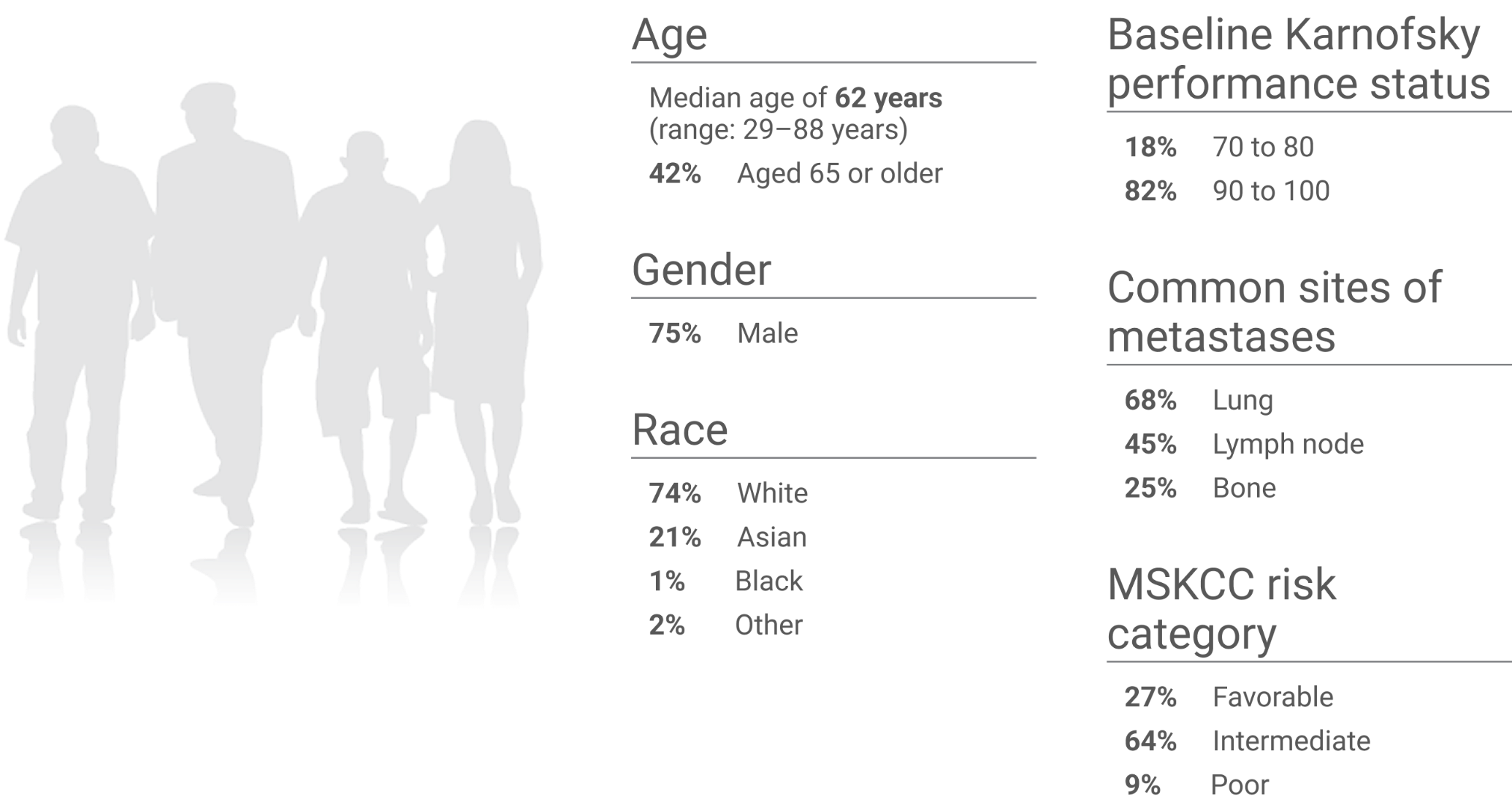
KEYNOTE-581/CLEAR trial: Baseline characteristics (N=1,069)1
Studied in the first-line setting across MSKCC risk groups
For the first-line treatment of adult patients with advanced RCC
KEYNOTE-581/CLEAR trial: Superior PFS with KEYTRUDA + LENVIMA vs sunitinib at protocol-specified interim analysis (HRa=0.39; 95% CI, 0.32–0.49; Pb<0.0001)
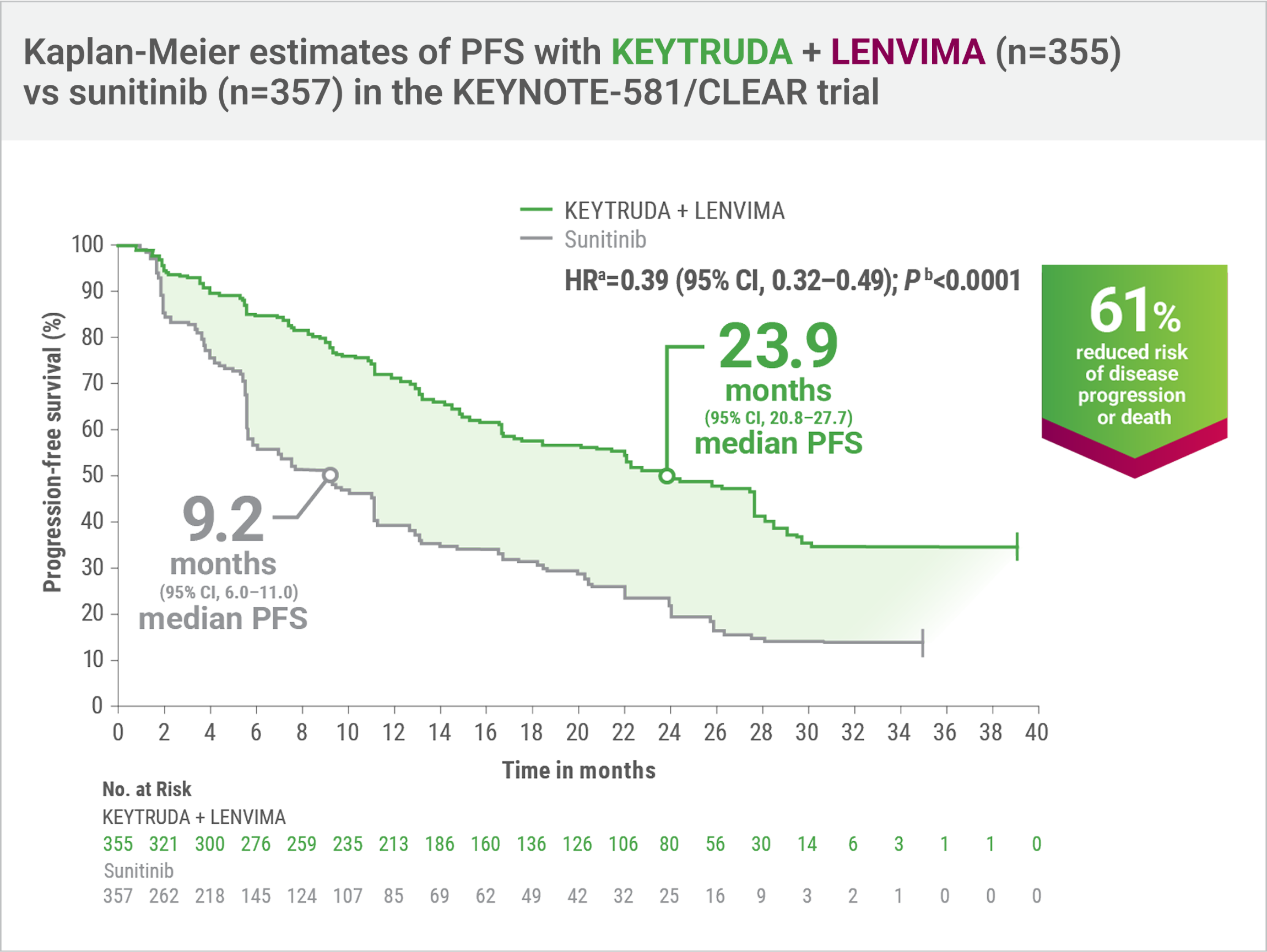
- Number of eventsc: 160/355 (45%) with KEYTRUDA + LENVIMA vs 205/357 (57%) with sunitinib; Progressive disease: 145/355 (41%) vs 196/357 (55%), respectively; Death: 15/355 (4%) vs 9/357 (3%), respectively
- Median PFS: 23.9 months (95% CI, 20.8–27.7) with KEYTRUDA + LENVIMA vs 9.2 months (95% CI, 6.0–11.0) with sunitinib
PFS and OS were major endpoints in the KEYNOTE-581/CLEAR trial.
Hazard ratio is based on a Cox Proportional Hazards Model. Stratified by geographic region and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic groups.
Two-sided P value based on stratified log-rank test.
Tumor assessments were based on RECIST v1.1; data cutoff date = 28 Aug 2020.
CI = confidence interval; HR = hazard ratio.
When considering treatment options for appropriate patients with advanced clear cell RCC, I recommend careful review of the clinical findings from the KEYNOTE-581/CLEAR trial. At the protocol-specified interim analysis, KEYTRUDA + LENVIMA demonstrated a 61% reduction in the risk of disease progression or death and a 2.5X greater median PFS vs sunitinib (23.9 months vs 9.2 months).
Thomas Hutson, DO, PharmD, PhD
Hem/Onc Division Chief
Professor of Medicine and Urology, UMC Cancer Center Director
Texas Tech University Health Science Center
Lubbock, TX
KEYNOTE-581/CLEAR trial
Exploratory subgroup analysis of PFS for KEYTRUDA + LENVIMA vs sunitinib in patients with advanced RCC based on selected baseline features2
LIMITATION: The KEYNOTE-581/CLEAR trial was not powered to detect differences in the treatment effect in these subgroups. No statistical testing was planned for this exploratory analysis, and no adjustment for multiplicity was made, therefore, no conclusions can be drawn.
This exploratory analysis of the KEYNOTE-581/CLEAR trial evaluated PFS for KEYTRUDA + LENVIMA vs sunitinib in patients with advanced RCC with or without specific baseline features, including baseline lung metastases, baseline bone metastases, baseline liver metastases, prior nephrectomy, sarcomatoid component by histology, IMDC risk group, and MSKCC risk group.a
PFS was assessed by IRC according to RECIST v1.1.
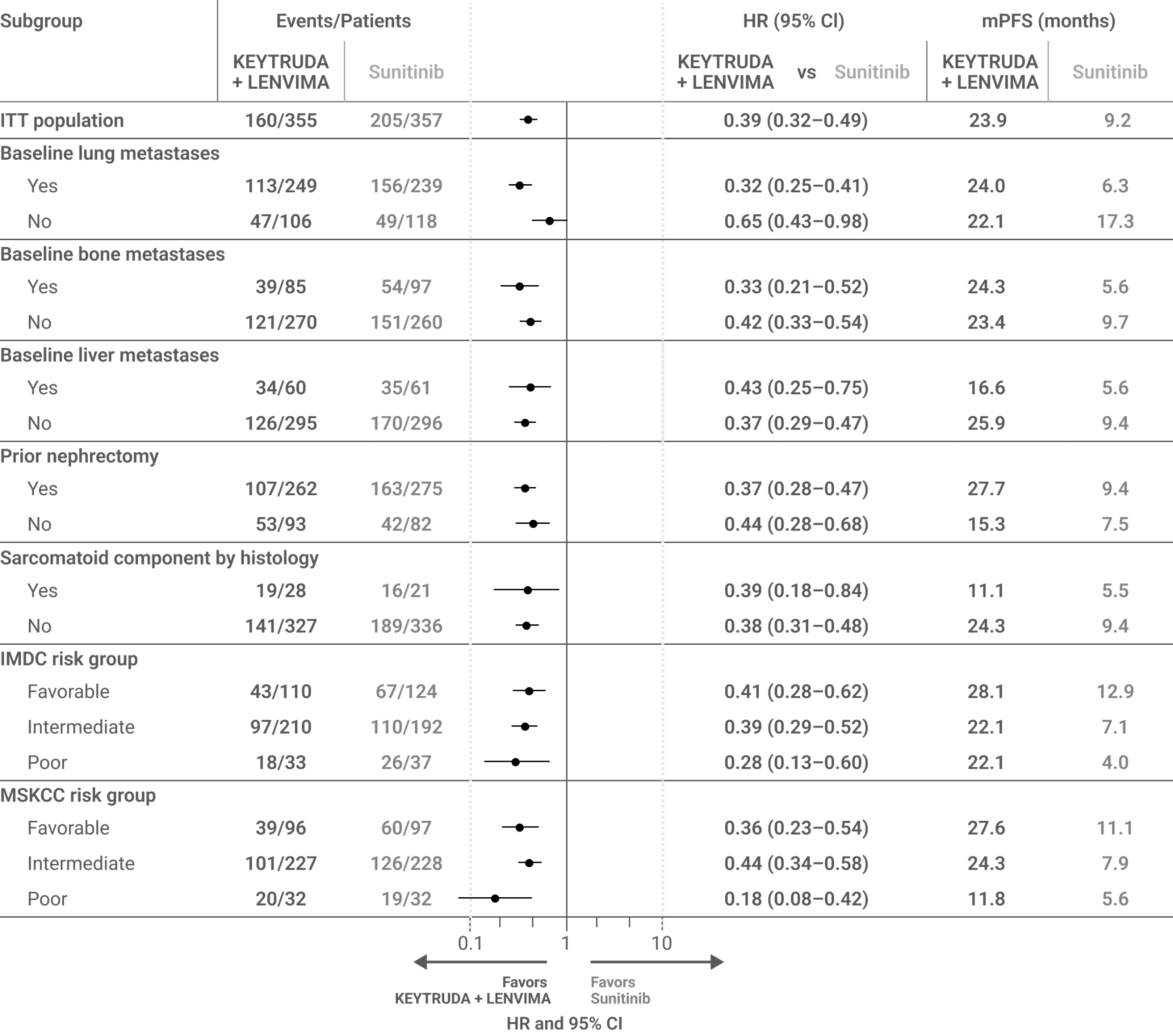
mPFS for the KEYTRUDA + LENVIMA and sunitinib arms was estimated using the Kaplan-Meier method; HR and 95% CIs comparing KEYTRUDA + LENVIMA with sunitinib were estimated by a stratified Cox model. If a stratification factor was itself a subgroup, this factor was removed from the stratified analysis.
IMDC = International Metastatic Renal Cell Carcinoma Database Consortium; ITT = intent to treat; mPFS = median progression-free survival.
Adapted from Grünwald V, Powles T, Eto M, et al. Front Oncol. 2023;13:1223282. doi:10.3389/fonc.2023.1223282, distributed under the terms of the Creative Commons Attribution License (CC BY).
KEYNOTE-581/CLEAR trial
Updated PFSa,b at final data cutoff3
LIMITATION: No statistical testing was planned for the updated PFS analysis, and therefore, no conclusions can be drawn.
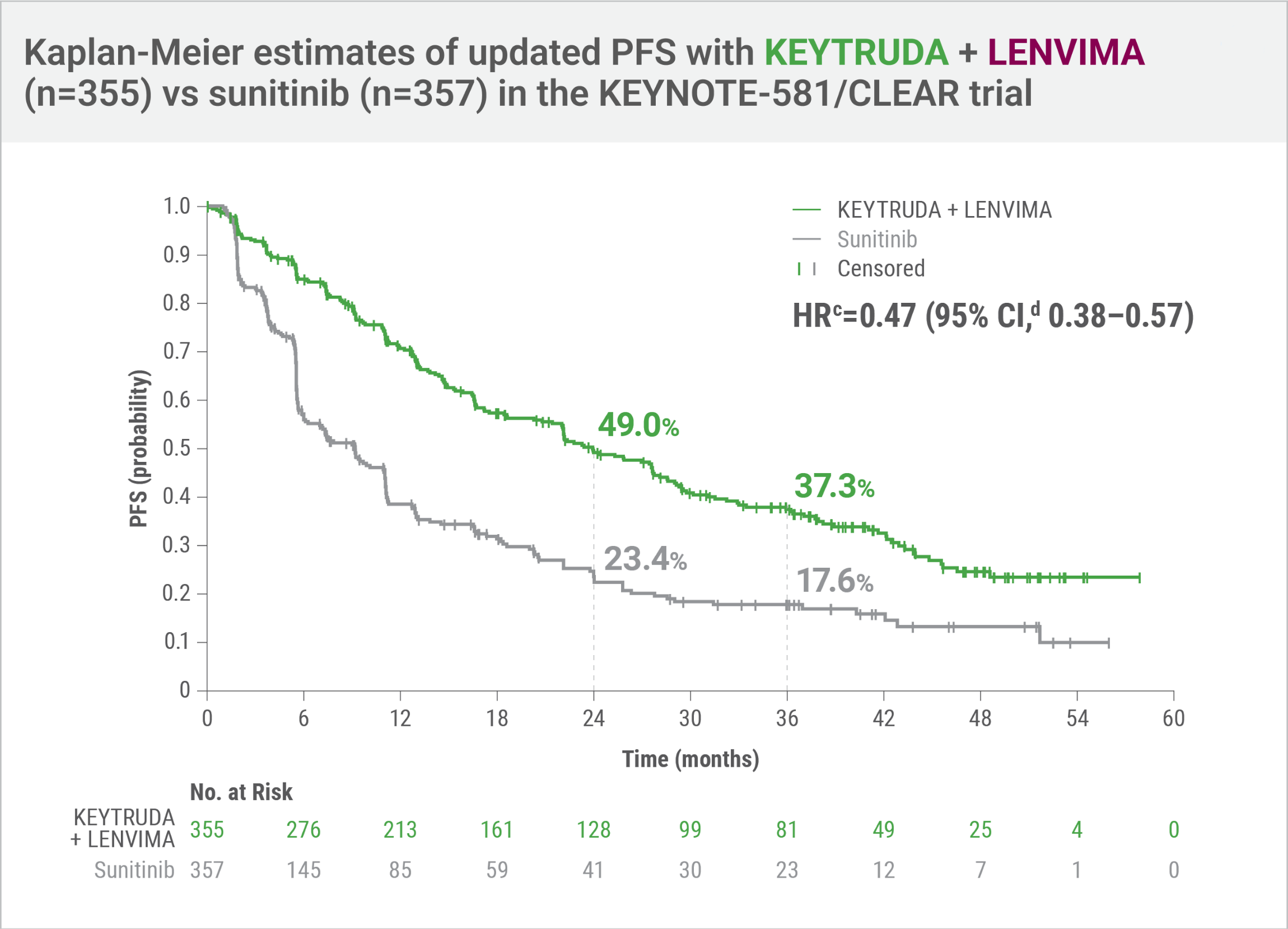
- HRc=0.47 (95% CI,d 0.38–0.57)
- Number of eventsb: 207/355 (58%) with KEYTRUDA + LENVIMA vs 214/357 (60%) with sunitinib
- Median PFS: 23.9 months (95% CI, 20.8–27.7) with KEYTRUDA + LENVIMA vs 9.2 months (95% CI, 6.0–11.0) with sunitinib
An updated PFS analysis was conducted when 304 deaths were observed based on the planned number of deaths for the prespecified final analysis.
Tumor assessments were based on independent imaging review per RECIST v1.1; updated OS cutoff date = 31 July 2022.
Hazard ratio is based on a Cox Proportional Hazards Model. Stratified by geographic region and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic groups.
95% CI estimation method using a generalized Brookmeyer and Crowley method.
For the first-line treatment of adult patients with advanced RCC
KEYNOTE-581/CLEAR trial: Durable overall survival with KEYTRUDA + LENVIMA vs sunitinib
Superior OS demonstrated with KEYTRUDA + LENVIMA vs sunitinib at protocol-specified interim analysis

- HRa=0.66; 95% CI, 0.49–0.88; Pb=0.0049
- Number of deathsc: 80/355 (23%) with KEYTRUDA + LENVIMA vs 101/357 (28%) with sunitinib
- Median OS was not reached (NR) in either arm: KEYTRUDA + LENVIMA (95% CI, 33.6–NR) and sunitinib (95% CI, NR–NR)
OS and PFS were major endpoints in the KEYNOTE-581/CLEAR trial.
For appropriate patients with advanced clear cell RCC, I consider overall survival data when selecting a treatment option. In the KEYNOTE-581/CLEAR trial, a 34% reduction in the risk of death was observed with KEYTRUDA + LENVIMA vs sunitinib at the protocol-specified interim analysis.
Nataliya Mar, MD
Associate Clinical Professor, Division of Hematology/Oncology
UCI Health
Orange, CA
Updated OSd,e at protocol-specified final analysis
This protocol-specified final analysis occurred after the interim analysis, which demonstrated the superiority of OS with KEYTRUDA + LENVIMA vs sunitinib. No statistical testing was planned for the protocol-specified final OS analysis.
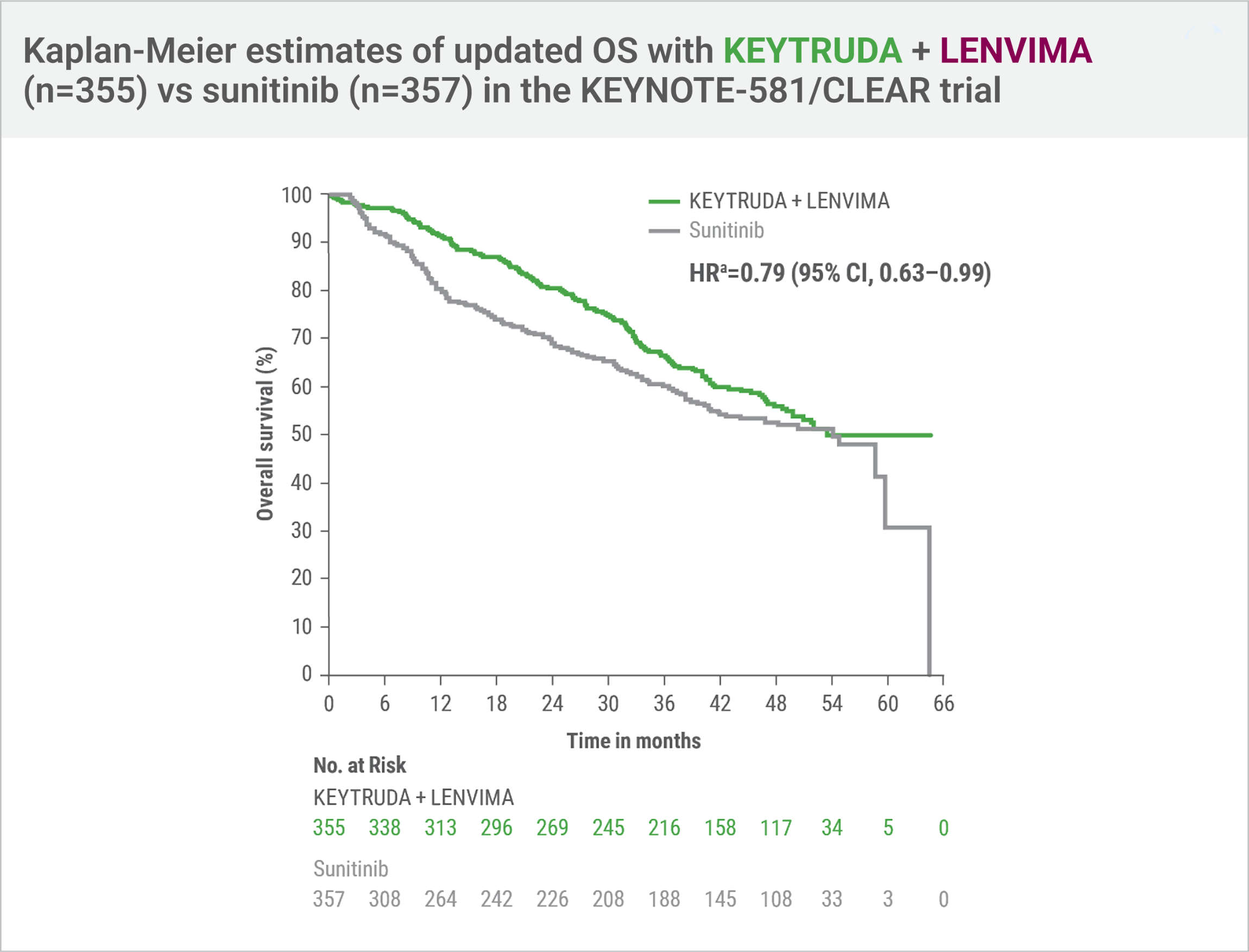
- HRa=0.79 (95% CI, 0.63–0.99)
- Number of deathse: 149/355 (42%) with KEYTRUDA + LENVIMA vs 159/357 (45%) with sunitinib
- Median OS: 53.7 months (95% CI, 48.7–NR) with KEYTRUDA + LENVIMA vs 54.3 months (95% CI, 40.9–NR) with sunitinib
Hazard ratio is based on a Cox Proportional Hazards Model. Stratified by geographic region and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic groups.
Two-sided P value based on stratified log-rank test.
Data cutoff date = 28 Aug 2020.
An updated OS analysis was conducted when 304 deaths were observed based on the planned number of deaths for the prespecified final analysis.
Updated OS cutoff date = 31 July 2022.
For the first-line treatment of adult patients with advanced RCC
KEYNOTE-581/CLEAR trial: A greater ORRa was demonstrated with KEYTRUDA + LENVIMA: 71% (95% CI, 66–76) vs 36% (95% CI, 31–41) with sunitinib (Pb<0.0001) at protocol-specified interim analysis
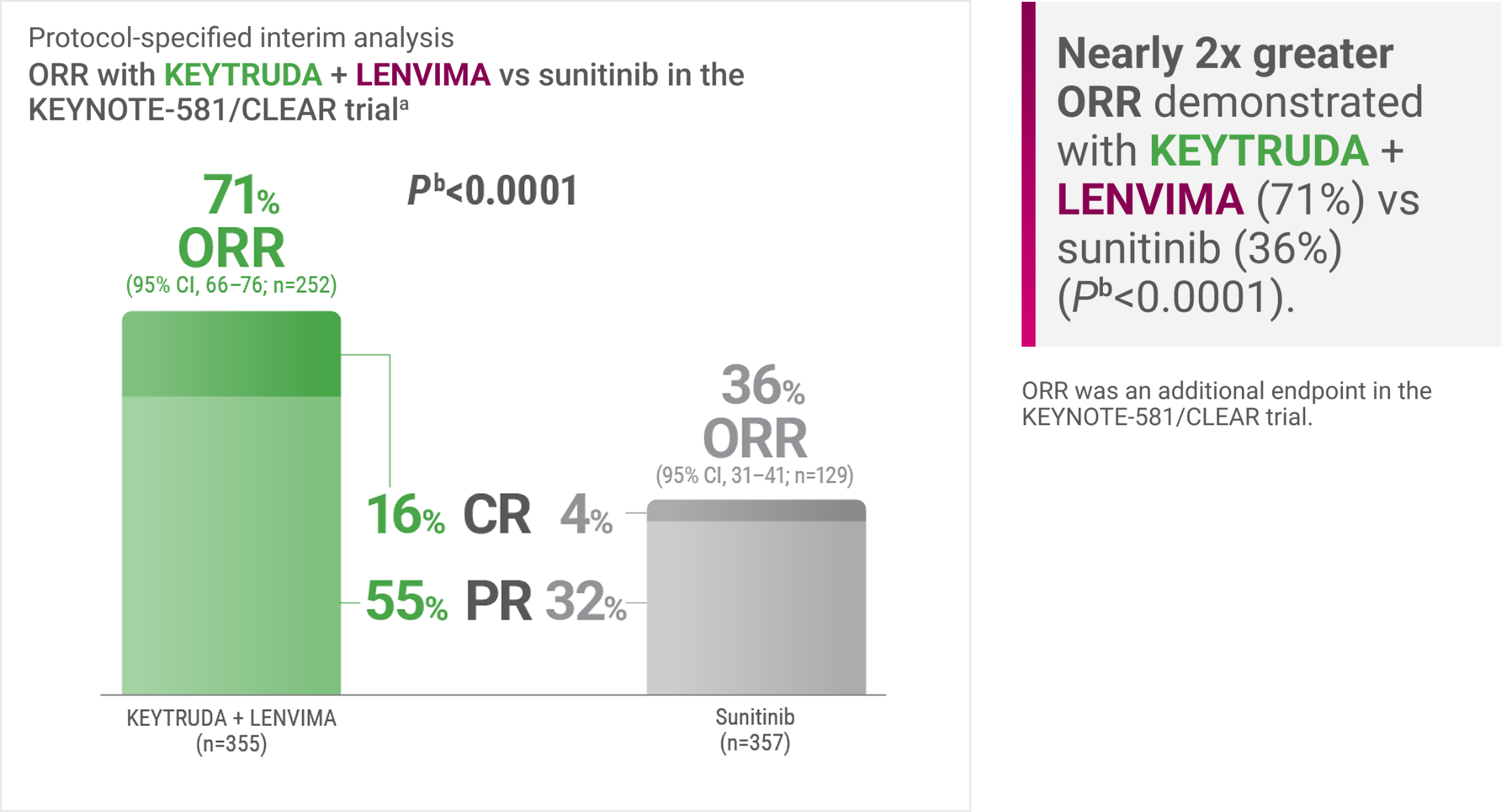
Tumor assessments were based on RECIST v1.1; only confirmed responses are included for ORR. Data cutoff date = 28 Aug 2020.
Two-sided P value based upon CMH test.
CMH = Cochran-Mantel-Haenszel; CR = complete response; ORR = objective response rate; PR = partial response.
ORR is one of several endpoints I review when selecting a treatment for appropriate patients with advanced clear cell RCC. In the KEYNOTE-581/CLEAR trial, KEYTRUDA + LENVIMA was associated with a nearly 2X greater ORR (71%) vs sunitinib (36%).
Raji Shameem, MD
Oncologist
Orlando Health
Orlando Regional Medical Center
Orlando, FL
KEYNOTE-581/CLEAR trial
Updated ORRa at final data cutoff3
LIMITATION: No statistical testing was planned for the updated ORR analysis, and therefore, no conclusions can be drawn.

Tumor assessments were based on independent imaging review per RECIST v1.1; only confirmed responses are included for ORR. Updated OS cutoff date = 31 July 2022.
95% CI is constructed using the method of normal approximation.
Includes 59 patients receiving KEYTRUDA + LENVIMA with a “near CR,” defined as a PR with ≥75% change from baseline in sum of target lesion diameters.
Includes 25 patients receiving sunitinib with a “near CR,” defined as a PR with ≥75% change from baseline in sum of target lesion diameter.
Disclaimer: FDA approval was based on the KEYNOTE-581/CLEAR trial for advanced RCC
KEYNOTE-B61: The first clinical trial in advanced or metastatic non-clear cell RCC (N=158) with IO + TKI data included in the FDA-approved labels4-7
A multicenter, single-arm trial that studied KEYTRUDA + LENVIMA in the first-line setting across IMDC risk groups and histologic subtypes, including papillary and chromophobe histologies
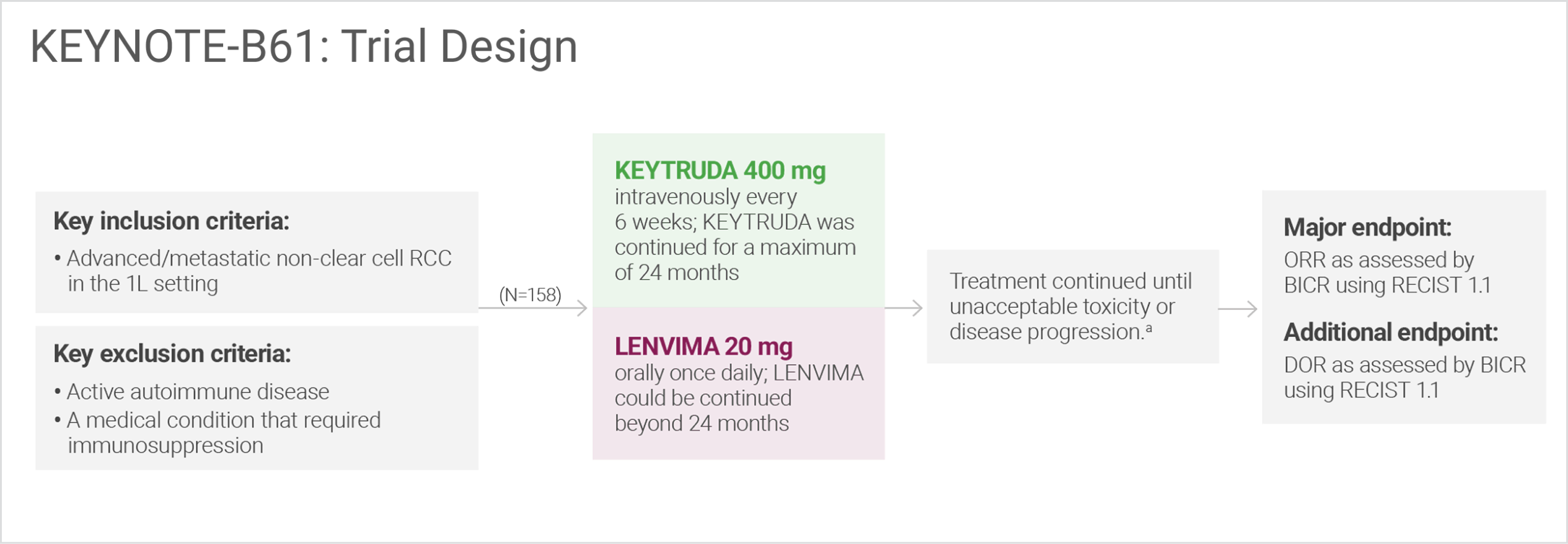
Administration of KEYTRUDA with LENVIMA was permitted beyond RECIST-defined disease progression if the patient was considered by the investigator to be deriving clinical benefit.
BICR = blinded independent central review; DOR = duration of response; IO = immunotherapy; TKI = tyrosine kinase inhibitor.
KEYNOTE-B61 evaluated KEYTRUDA + LENVIMA in a cohort of 158 patients with advanced non-clear cell RCC. I encourage careful review of the KEYNOTE-B61 data which evaluated patients across multiple histologic subtypes.
Thomas Hutson, DO, PharmD, PhD
Hem/Onc Division Chief
Professor of Medicine and Urology, UMC Cancer Center Director
Texas Tech University Health Science Center
Lubbock, TX
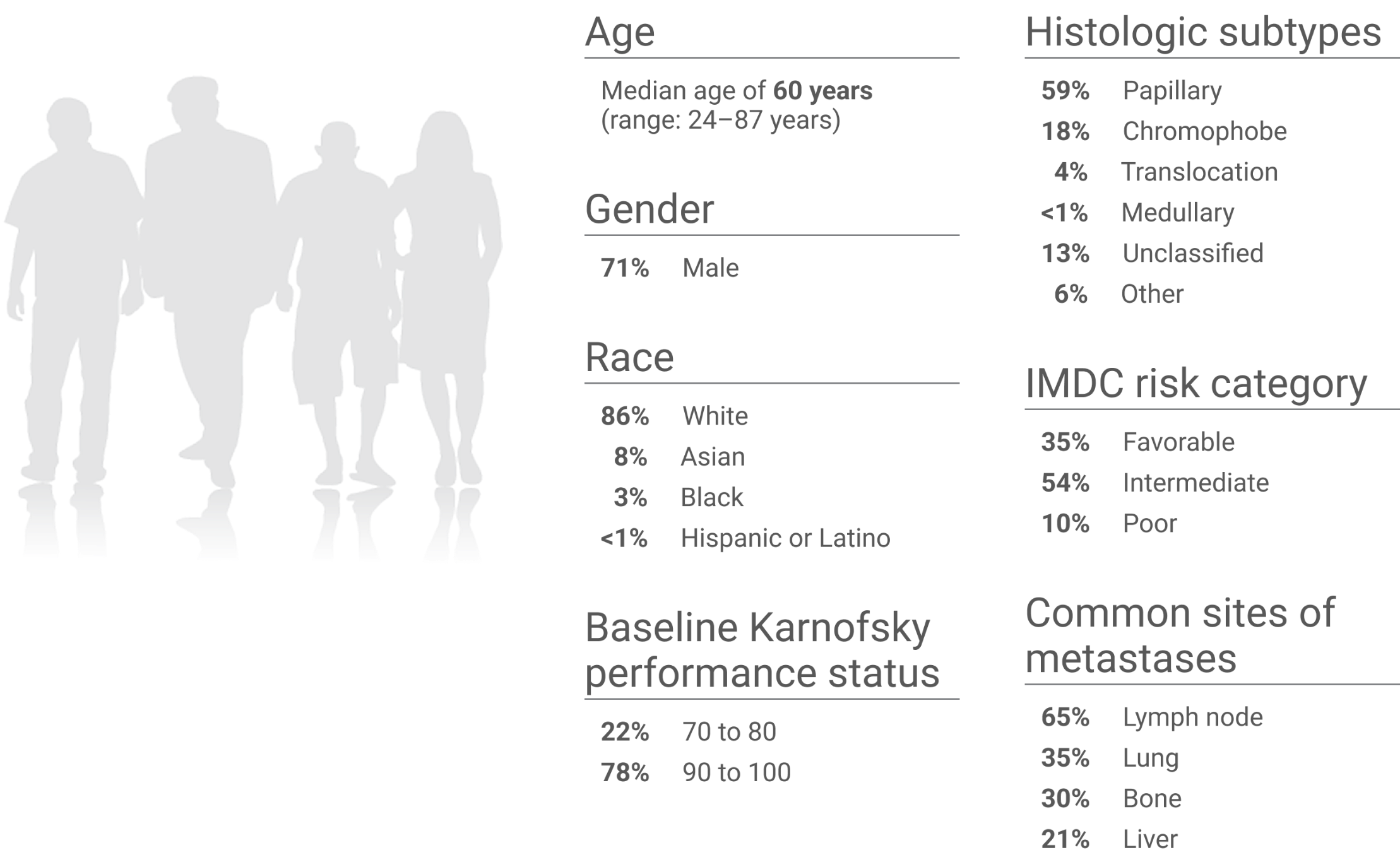
KEYNOTE-B61 trial: Baseline characteristics (N=158)
Studied in the first-line setting across IMDC risk groups and histologic subtypes
For the first-line treatment of adult patients with advanced RCC
In the single-arm KEYNOTE-B61 trial: Half of patients with advanced or metastatic non-clear cell RCC had a confirmed objective response with KEYTRUDA + LENVIMA
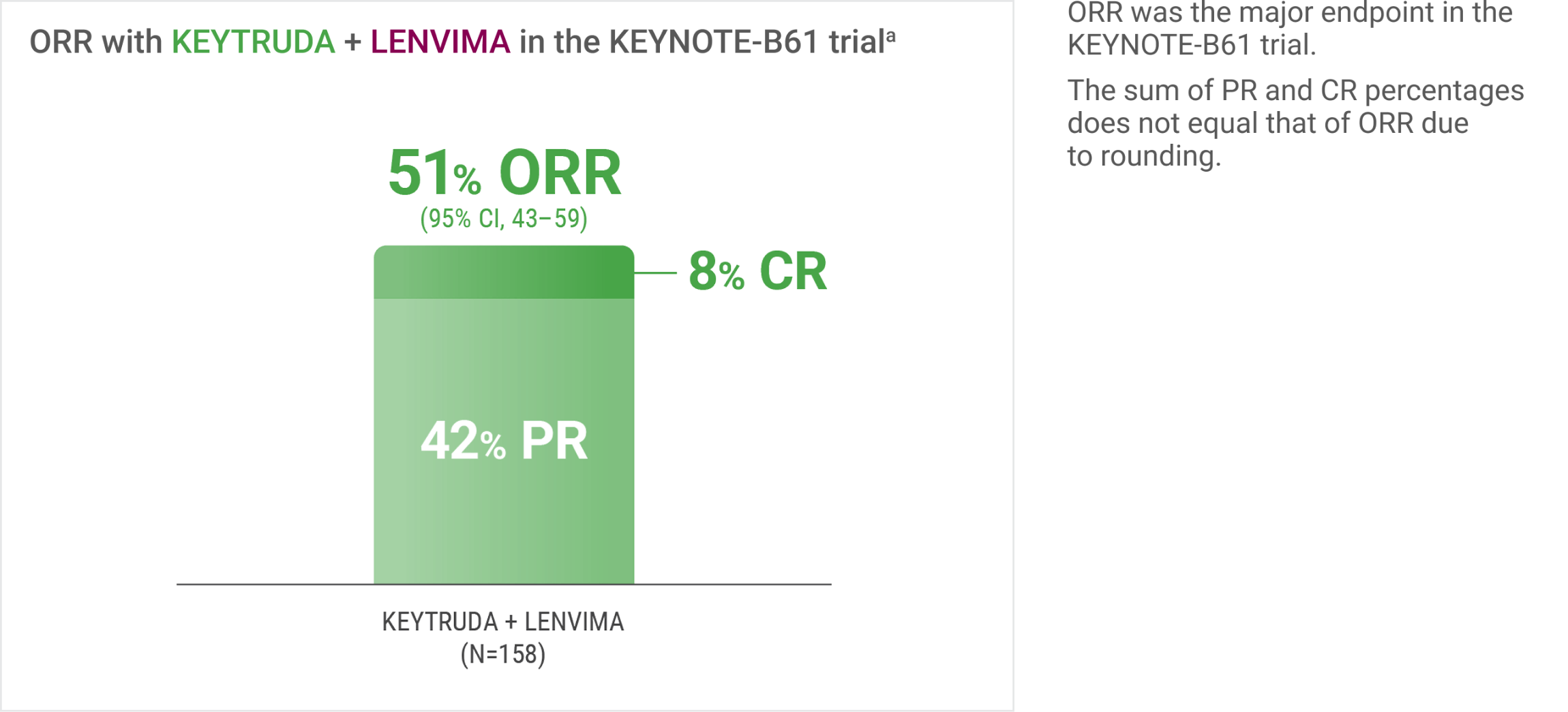
ORR was assessed by BICR using RECIST 1.1.
The data from KEYNOTE-B61 helps inform treatment decisions for appropriate patients with advanced non-clear cell RCC. Patients with advanced or metastatic non-clear cell RCC demonstrated objective response results, with 51% of those treated with KEYTRUDA + LENVIMA having a confirmed objective response.
Raji Shameem, MD
Oncologist
Orlando Health
Orlando Regional Medical Center
Orlando, FL
For the first-line treatment of adult patients with advanced RCC
In the single-arm KEYNOTE-B61 trial: Confirmed objective response was observed with KEYTRUDA + LENVIMA across multiple histologic subtypes
LIMITATION: No statistical testing was conducted to compare histologic subtypes in this single-arm study. This descriptive analysis is limited by the small sample sizes of each histologic subtype. Therefore, no conclusions can be drawn.
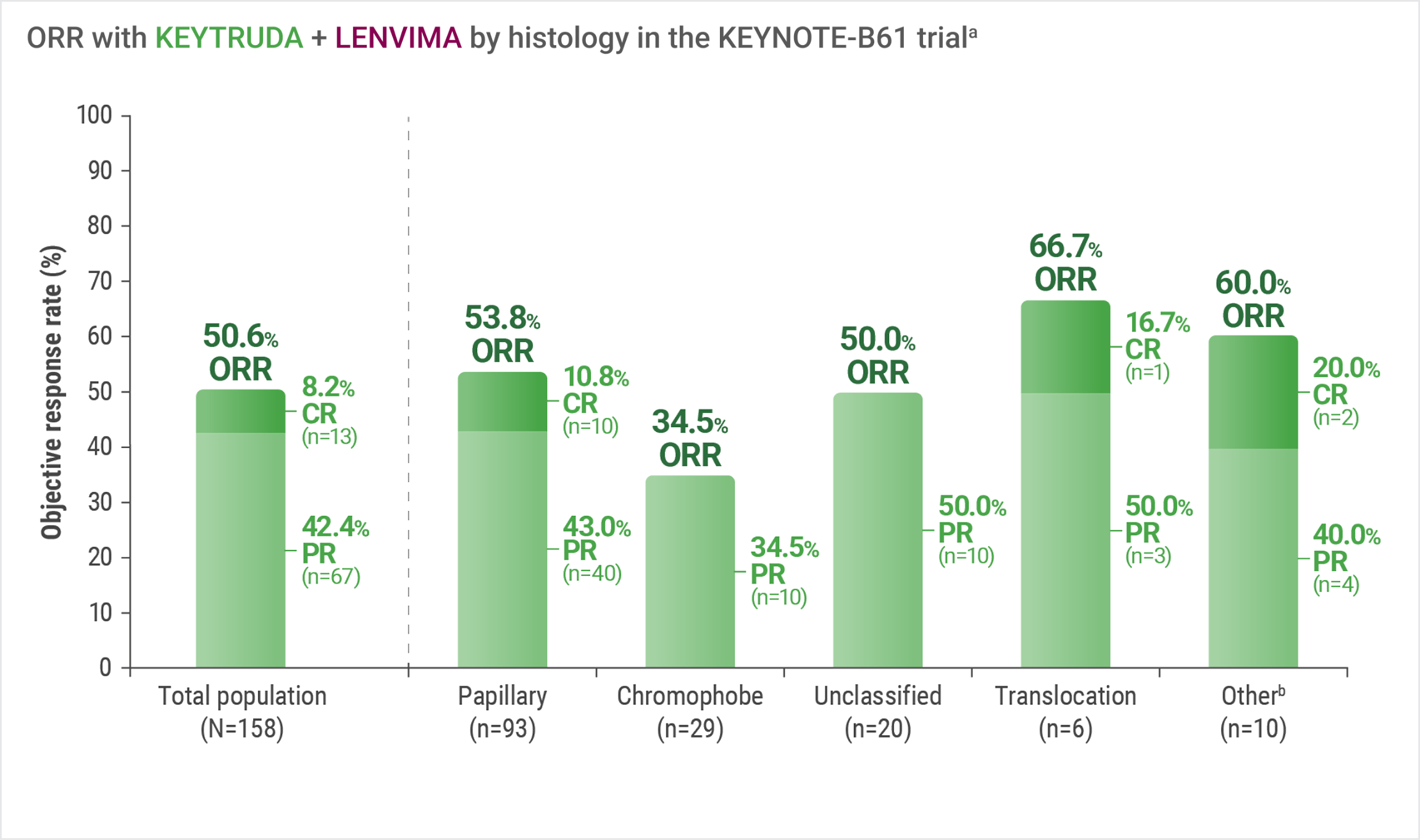
Assessed by BICR using RECIST 1.1.
Includes other non-clear cell histologic subtypes.
For the first-line treatment of adult patients with advanced RCC
In the single-arm KEYNOTE-B61 trial: A median DORa,b of 19.5 months (range: 1.5+ to 23.5+) was observed with KEYTRUDA + LENVIMA

DOR was an additional endpoint in the KEYNOTE-B61 trial.
Based on Kaplan-Meier estimates.
“+” denotes ongoing response.
I choose KEYTRUDA + LENVIMA for my appropriate patients with advanced non-clear cell RCC. In the KEYNOTE-B61 trial, a median duration of response of 19.5 months was reported with KEYTRUDA + LENVIMA, which is one endpoint I consider when making a treatment decision.
Nataliya Mar, MD
Associate Clinical Professor, Division of Hematology/Oncology
UCI Health
Orange, CA
For the first-line treatment of adult patients with advanced RCC
Adverse reactions (ARs) in the KEYNOTE-581/CLEAR trial
The safety of KEYTRUDA + LENVIMA was investigated in the KEYNOTE-581/CLEAR trial in patients treated with KEYTRUDA + LENVIMA (n=352) compared to sunitinib (n=340) at the protocol-specified interim analysis.
The median duration of exposure to KEYTRUDA + LENVIMA was 17 months (range: 0.1 to 39).
Fatal adverse reactions occurred in 4.3% of patients treated with KEYTRUDA + LENVIMA, including cardio-respiratory arrest (0.9%), sepsis (0.9%), and one case (0.3%) each of:
- Arrhythmia
- Autoimmune hepatitis
- Dyspnea
- Hypertensive crisis
- Increased blood creatinine
- Multiple organ dysfunction syndrome
- Myasthenic syndrome
- Myocarditis
- Nephritis
- Pneumonitis
- Ruptured aneurysm
- Subarachnoid hemorrhage
Serious adverse reactions occurred in 51% of patients receiving KEYTRUDA + LENVIMA.
Serious adverse reactions in ≥2% of patients receiving KEYTRUDA + LENVIMA were:
- Hemorrhagic events (5%)
- Diarrhea (4%)
- Hypertension (3%)
- Myocardial infarction (3%)
- Pneumonitis (3%)
- Vomiting (3%)
- Acute kidney injury (2%)
- Adrenal insufficiency (2%)
- Dyspnea (2%)
- Pneumonia (2%)
| Permanent Discontinuation (%) |
Dose Interruption (%) |
Dose Reduction (%) |
|
|---|---|---|---|
| KEYTRUDA, LENVIMA, or both | 37 | 78 | — |
| KEYTRUDA + LENVIMA | 13 | 39 | — |
| KEYTRUDA only | 29 | 55 | — |
| LENVIMA only | 26 | 73 | 69 |
| KEYTRUDA, LENVIMA, or both | Permanent Discontinuation (%)37 | Dose Interruption (%)78 | Dose Reduction (%)— |
| KEYTRUDA + LENVIMA | Permanent Discontinuation (%)13 | Dose Interruption (%)39 | Dose Reduction (%)— |
| KEYTRUDA only | Permanent Discontinuation (%)29 | Dose Interruption (%)55 | Dose Reduction (%)— |
| LENVIMA only | Permanent Discontinuation (%)26 | Dose Interruption (%)73 | Dose Reduction (%)69 |
- No dose reduction for KEYTRUDA is recommended.
The most common (≥2%) adverse reactions that resulted in permanent discontinuation of KEYTRUDA, LENVIMA, or both
- Pneumonitis (3%)
- Myocardial infarction (3%)
- Hepatotoxicity (3%)
- Acute kidney injury (3%)
- Rash (3%)
- Diarrhea (2%)
Most common adverse reactions that resulted in dose reduction or interruption in the KEYNOTE-581/CLEAR trial
Most common (≥3%) adverse reactions in patients receiving KEYTRUDA + LENVIMA that resulted in interruption of KEYTRUDA
- Diarrhea (10%)
- Hepatotoxicity (8%)
- Fatigue (7%)
- Lipase increased (5%)
- Amylase increased (4%)
- Musculoskeletal pain (3%)
- Hypertension (3%)
- Rash (3%)
- Acute kidney injury (3%)
- Decreased appetite (3%)
Most common (≥5%) adverse reactions in patients receiving KEYTRUDA + LENVIMA that resulted in dose reduction or interruption of LENVIMA
- Diarrhea (26%)
- Fatigue (18%)
- Hypertension (17%)
- Proteinuria (13%)
- Decreased appetite (12%)
- Palmar-plantar erythrodysesthesia (11%)
- Nausea (9%)
- Stomatitis (9%)
- Musculoskeletal pain (8%)
- Rash (8%)
- Increased lipase (7%)
- Abdominal pain (6%)
- Vomiting (6%)
- Increased ALT (5%)
- Increased amylase (5%)
Adverse reactions that occurred in ≥20% of patients receiving KEYTRUDA + LENVIMA in the KEYNOTE-581/CLEAR trial
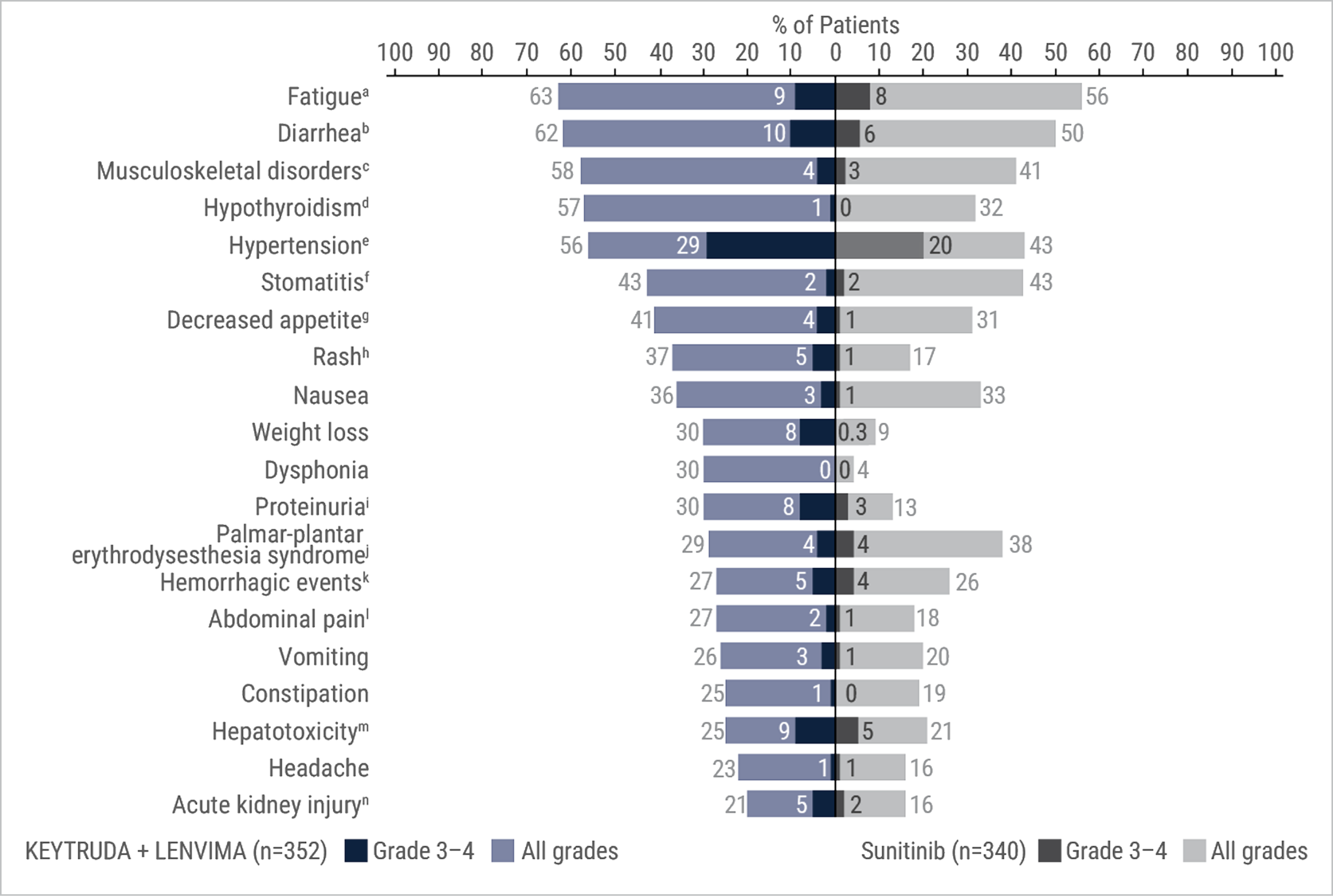
- Fifteen percent (15%) of patients treated with KEYTRUDA + LENVIMA received an oral prednisone equivalent to ≥40 mg daily for an immune-mediated adverse reaction.
- Clinically relevant adverse reactions (<20%) that occurred in patients receiving KEYTRUDA + LENVIMA were myocardial infarction (3%) and angina pectoris (1%).
- Grade 3 and 4 increased ALT or AST was seen in 9% of patients. Grade ≥2 increased ALT or AST was reported in 64 (18%) patients, of whom 20 (31%) received ≥40 mg daily oral prednisone equivalent. Recurrence of Grade ≥2 increased ALT or AST was observed on rechallenge in 3 patients receiving LENVIMA, in 10 patients receiving both KEYTRUDA and LENVIMA (n=38), and was not observed on rechallenge with KEYTRUDA alone (n=3).
Includes asthenia, fatigue, lethargy, malaise.
Includes diarrhea, gastroenteritis.
Includes arthralgia, arthritis, back pain, bone pain, breast pain, musculoskeletal chest pain, musculoskeletal discomfort, musculoskeletal pain, musculoskeletal stiffness, myalgia, neck pain, non-cardiac chest pain, pain in extremity, pain in jaw.
Includes hypothyroidism, increased blood thyroid stimulating hormone, secondary hypothyroidism.
Includes essential hypertension, increased blood pressure, increased diastolic blood pressure, hypertension, hypertensive crisis, hypertensive retinopathy, labile blood pressure.
Includes aphthous ulcer, gingival pain, glossitis, glossodynia, mouth ulceration, mucosal inflammation, oral discomfort, oral mucosal blistering, oral pain, oropharyngeal pain, pharyngeal inflammation, stomatitis.
Includes decreased appetite, early satiety.
Includes genital rash, infusion site rash, penile rash, perineal rash, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular.
Includes hemoglobinuria, nephrotic syndrome, proteinuria.
Includes palmar erythema, palmar-plantar erythrodysesthesia syndrome, plantar erythema.
Includes all hemorrhage terms. Hemorrhage terms that occurred in 1 or more subjects in either treatment group include anal hemorrhage, aneurysm ruptured, blood blister, blood loss anemia, blood urine present, catheter site hematoma, cerebral microhemorrhage, conjunctival hemorrhage, contusion, diarrhea hemorrhagic, disseminated intravascular coagulation, ecchymosis, epistaxis, eye hemorrhage, gastric hemorrhage, gastritis hemorrhagic, gingival bleeding, hemorrhage urinary tract, hemothorax, hematemesis, hematoma, hematochezia, hematuria, hemoptysis, hemorrhoidal hemorrhage, increased tendency to bruise, injection site hematoma, injection site hemorrhage, intra-abdominal hemorrhage, lower gastrointestinal hemorrhage, Mallory-Weiss syndrome, melaena, petechiae, rectal hemorrhage, renal hemorrhage, retroperitoneal hemorrhage, small intestinal hemorrhage, splinter hemorrhages, subcutaneous hematoma, subdural hematoma, subarachnoid hemorrhage, thrombotic thrombocytopenic purpura, tumor hemorrhage, traumatic hematoma, upper gastrointestinal hemorrhage.
Includes abdominal discomfort, abdominal pain, abdominal rigidity, abdominal tenderness, epigastric discomfort, lower abdominal pain, upper abdominal pain.
Includes alanine aminotransferase increased, aspartate aminotransferase increased, blood bilirubin increased, drug-induced liver injury, hepatic enzyme increased, hepatic failure, hepatic function abnormal, hepatocellular injury, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, immune-mediated hepatitis, liver function test increased, liver injury, transaminases increased, gammaglutamyltransferase increased.
Includes acute kidney injury, azotemia, blood creatinine increased, creatinine renal clearance decreased, hypercreatininemia, renal failure, renal impairment, oliguria, glomerular filtration rate decreased, nephropathy toxic.
ALT = alanine aminotransferase; AST = aspartate aminotransferase.
Post hoc analysis: Median time to first onset of select adverse reactions with KEYTRUDA + LENVIMA in KEYNOTE-581/CLEAR (n=352)8
LIMITATION: This is a post hoc analysis based on data from KEYNOTE-581/CLEAR. No formal statistical testing was planned and, therefore, no conclusions can be drawn.
- As this information is descriptive only, it may not be reflective of clinical practice; it should not replace physician judgement and evaluation of a potential adverse reaction should it occur.
- Health care professionals should monitor and evaluate patients for the presence of potential adverse reactions throughout treatment with KEYTRUDA, in combination with LENVIMA, and following discontinuation.
- Immune-mediated adverse reactions can occur at any time after starting treatment with a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
- Data does not represent a complete list of each adverse reaction that occurred during the CLEAR trial.
- The interquartile range (Q1:Q3) represents the time to onset (the earliest treatment-emergent adverse reaction [AR] start date) for the AR for the middle 50% of patients who experienced that AR from quartile 1 to quartile 3.
- ARs were chosen based on frequency of occurrence (in ≥30% of patients). ARs could have occurred while receiving LENVIMA and/or KEYTRUDA or within the protocol-defined follow-up period of 30 days after the patient’s last dose. ARs were recorded until the end of the follow-up period or until resolution, whichever came first. Grading of ARs was performed according to Common Terminology Criteria for Adverse Events v4.03.
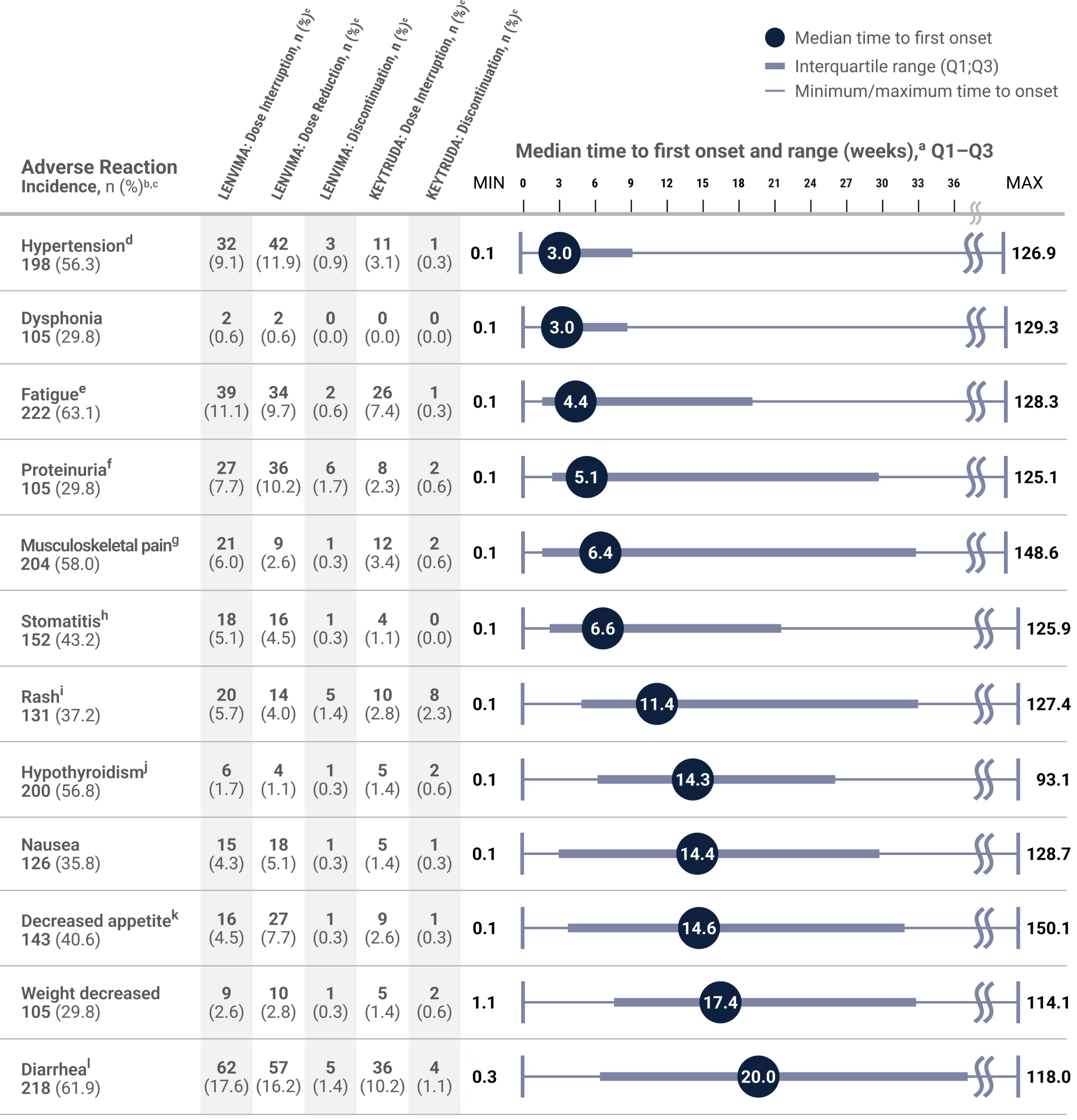
Chart adapted with permission from Motzer R, George S, Merchan JR, et al. Characterization and management of adverse reactions from the CLEAR study in advanced renal cell carcinoma treated with lenvatinib plus pembrolizumab. Oncologist. 2023;28(6):501–509. doi:10.1093/oncolo/oyac269
Median time to first onset in patients who experienced the adverse reaction. Gray boxes represent Q1–Q3. Lines represent the range.
Any grade.
Percentages are based on the safety population of the KEYTRUDA + LENVIMA group (n=352). The safety population included all patients who received at least 1 dose of any study drug.
Includes essential hypertension, increased blood pressure, increased diastolic blood pressure, hypertension, hypertensive crisis, hypertensive retinopathy, and labile blood pressure.
Includes fatigue, asthenia, malaise, and lethargy.
Includes hemoglobinuria, nephrotic syndrome, and proteinuria.
Includes arthralgia, arthritis, back pain, bone pain, breast pain, musculoskeletal chest pain, musculoskeletal discomfort, musculoskeletal pain, musculoskeletal stiffness, myalgia, neck pain, noncardiac chest pain, pain in extremity, and pain in jaw.
Aphthous ulcer, gingival pain, glossitis, glossodynia, mouth ulceration, mucosal inflammation, oral discomfort, oral mucosal blistering, oral pain, oropharyngeal pain, pharyngeal inflammation, and stomatitis.
Includes genital rash, infusion site rash, penile rash, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, and rash pustular.
Includes hypothyroidism, increased blood thyroid-stimulating hormone, and secondary hypothyroidism.
Includes decreased appetite and early satiety.
Includes diarrhea and gastroenteritis.
MAX = maximum; MIN = minimum; PD-1 = programmed death receptor-1; PD-L1 = programmed death ligand 1; Q1 = first quartile; Q3 = third quartile.
References: 1. Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384(14):1289-1300. doi:10.1056/NEJMoa2035716 2. Grünwald V, Powles T, Eto M, et al. Phase 3 CLEAR study in patients with advanced renal cell carcinoma: outcomes in subgroups for the lenvatinib-plus-pembrolizumab and sunitinib arms. Front Oncol. 2023;13:1223282. doi:10.3389/fonc.2023.1223282 3. Motzer R, Porta C, Eto M, et al. Lenvatinib plus pembrolizumab versus sunitinib in first-line treatment of advanced renal cell carcinoma: final prespecified overall survival analysis of CLEAR, a phase III study. J Clin Oncol. 2024;42(11):1222-1228. doi:10.1200/JCO.23.01569 4. Stellato M, Buti S, Maruzzo M, et al. Pembrolizumab plus axitinib for metastatic papillary and chromophobe renal cell carcinoma: NEMESIA (Non Clear MEtaStatic Renal Cell Carcinoma Pembrolizumab Axitinib) study, a subgroup analysis of I-RARE observational study (Meet-URO 23a). Int J Mol Sci. 2023;24(2):1096. doi:org/10.3390/iims24021096 5. Lee CH, Voss MH, Carlo MI, et al. Phase II trial of cabozantinib plus nivolumab in patients with non-clear-cell renal cell carcinoma and genomic correlates. J Clin Oncol. 2022;40(21):2333-2341. doi:10.1200/JCO.21.01944 6. McGregor BA, McKay RR, Braun DA, et al. Results of a multicenter phase II study of atezolizumab and bevacizumab for patients with metastatic renal cell carcinoma with variant histology and/or sarcomatoid features. J Clin Oncol. 2020;38(1):63-70. doi:10.1200/JCO.19.01882 7. Dong P, Wei W, Jiang L, et al. Anlotinib combined with sintilimab as first-line treatment in patients with advanced non-clear cell renal cell carcinoma: preliminary results from an exploratory prospective clinical study. J Clin Oncol. 2023;41(suppl 16):e16546–e16546. doi:10.1200/JCO.2023.41.16_suppl.e16546 8. Motzer R, George S, Merchan JR, et al. Characterization and management of adverse reactions from the CLEAR study in advanced renal cell carcinoma treated with lenvatinib plus pembrolizumab. The Oncologist. 2023;28(6):501-509. doi:10.1093/oncolo/oyac269